.
Modern cell research is increasingly relying on single cell analysis. Whether it is cell analysis in microbiology, immune-oncology or drug discovery processes, gentle single cell dissociation from their matrix is a prerequisite for proper single cell analysis.
OLS, your ideal cell research partner, is proud to present to you TIGR Tissue Grinder and Dissociator, the semi-automated, enzyme-free, fast and reproducible approach of generating pure and individual single cells from tissue samples.

TIGR Tissue & Dissociator in the new OLS Lab in Bremen
Highlights
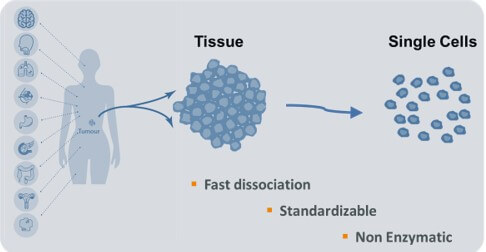
- Enzyme-free, standardized approach enables sophisticated applications
- 5 to 15x faster dissociation
- Integrated workflow for minimal contamination
- Easy to use software and customizable protocols
- Patented technology (EU, US, CN)
- Gentle sample preparation
- Easy to use software
TIGR is compatible with standard cell culture consumables 50 ml centrifuge tubes and standard cell strainer for mechanically, non-enzymatic and parallel processing of tissue samples in a reduced processing time.
TIGR is composed of a benchtop instrument and the grinding tube, a sterile disposable with the integrated core unit and cell strainer. The two-part core unit, consisting of rotor and stator inserts is equipped with counter rotating grinding teeth.
One of the main advantages of TIGR is its flexibility. The process parameters time, direction of rotation, acceleration, speed and the number of process steps can be adjusted by the researcher. Indeed, this allows creating an-experiment specific protocol for every application.
Another major advantage is saving time. The dissociation by TIGR was completed in less than two minutes. This is substantially less when benchmarked against other enzyme based technologies, methods that take 60 minutes, especially when multiple samples are processed in parallel.
Principle
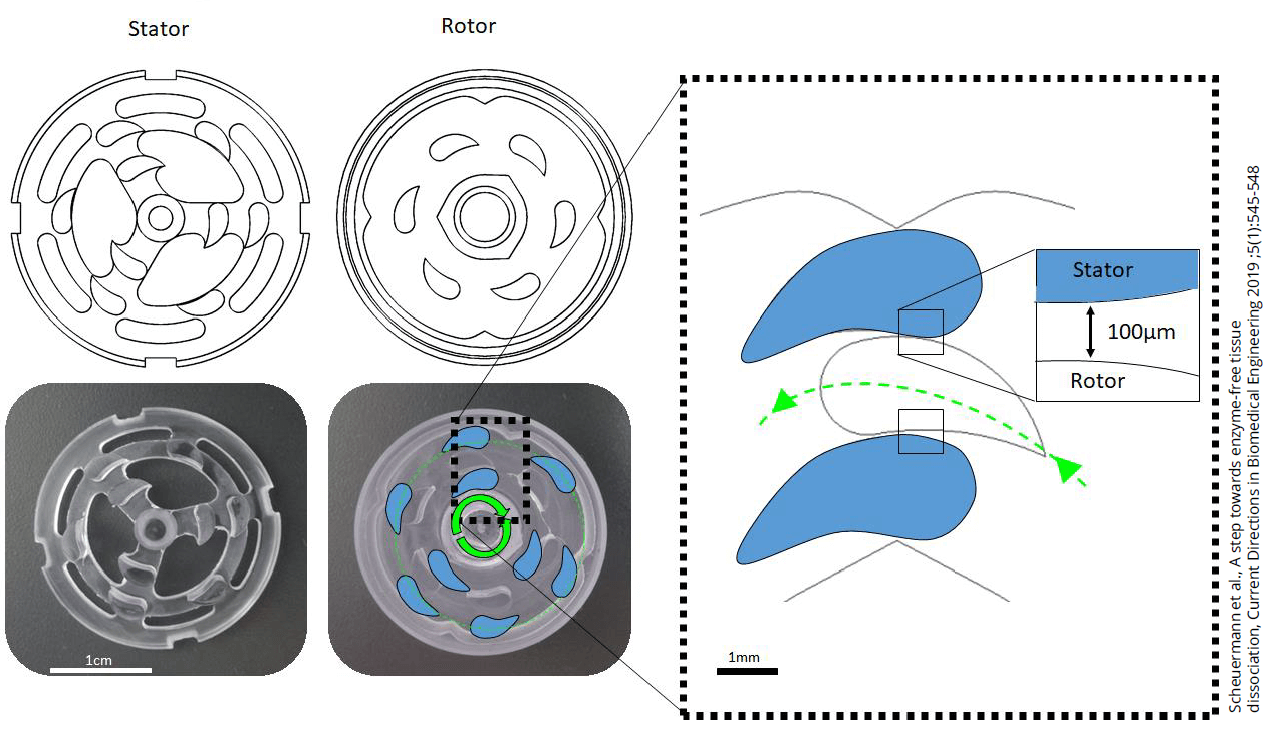 Underlying principle of the TIGR Tissue Grinder and Dissociator
Underlying principle of the TIGR Tissue Grinder and DissociatorThe grinding unit interlocks with rows of teeth that can be moved against each other. The free rotation of the grinding teeth draws the tissue sample into the free space applying adequate amounts of shearing and milling force to gently isolate single cell from the tissue. The space between the grinding teeth and their fin-shaped geometry allows efficient extraction of viable single cells. The stator is the part that does not move, while the rotor is moved bidirectionally, depending on your customizable programming, allowing for user-defined and -controlled dissociation of the cell tissue.
Established Protocols
| Using default protocol | With protocol optimization |
|---|---|
| Liver | Skin* |
| Lung | FFPE* |
| Heart | Brain |
| Kidney | Urinary bladder |
| Spleen | Thyroid |
| Stomach | Tonsil |
| Colon | Prostate |
| Pancreas | Endometrium |
| Lymph node | Ovary |
| Rectum |
The tissue types found in the left column already have established protocols provided alongside the TIGR Tissue Grinder & Dissociator, as well as a good database from multiple users in cell research. Because of this, any new user can rely on these protocols without needing to make major adjustments, unless for special requirements in the downstream application. The right column shows tissue types that have been proven to be compatible with the TIGR Tissue Grinder, but might need protocol optimization on a case-to-case basis. We are happy to assist in the establishment and testing of new protocols and provide data on these tissue types whereever available. *Skin and FFPE will benefit from the addition of dissociation enzymes during the grinding process.
Many more tissue types are currently in testing steps. If you have tissue not listed in this table, please let us know – we are happy to conduct tests and generate data in cooperation with end users.
Applications
- 3D Cell Culture
- Tissue Models – Spheroids, Organoids, Tumoroids
- Single Cell Counting
- Isolation of Primary Cells
- Cancer Cell Line Development
- Flow Cytometry
Workflow
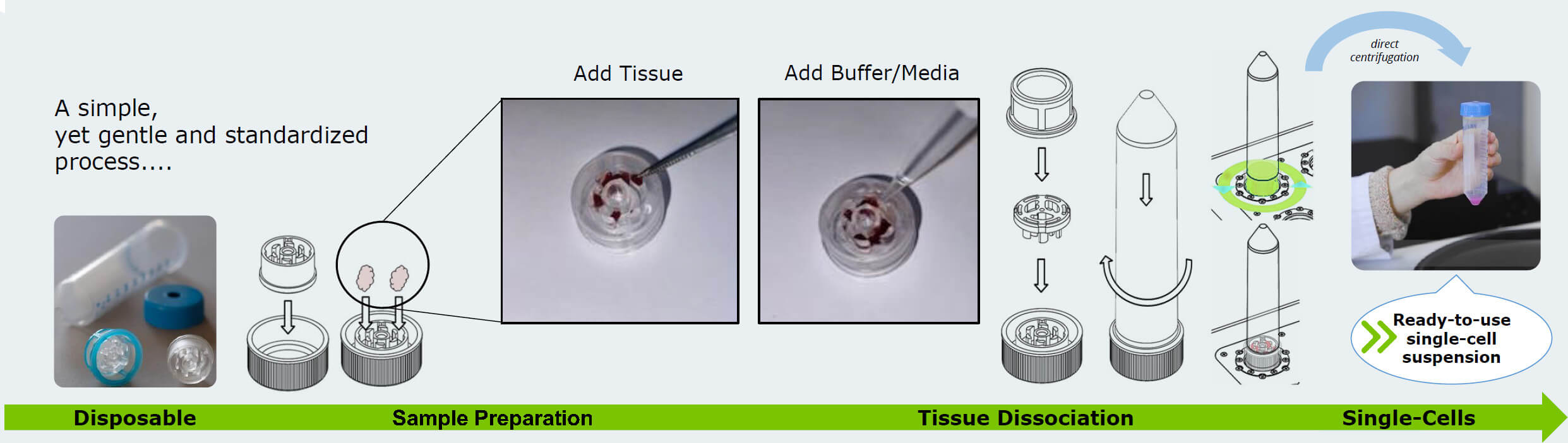
The tissue grinding and dissociation with the TIGR follows a simple and standardized workflow. In sterile conditions, the tissue sample is placed between rotor and stator, alongside with some liquid for the cells to be suspended in. Then the cell strainer and the tube are placed over the grinding compartment. From here on out, the tube can be taken out of the sterile space. The tube has to be placed on a TIGR slot with the upside down, so the rotor can connect to the slot’s rotation gear. After using the TIGR software to execute the grinding protocol, the tube can be taken off the instrument and flipped right side up, so that the suspension will pass the cell strainer. The final step to receive a ready-to-use single-cell suspension is centrifugation and resuspension. This quick and easy-to-follow workflow is all it needs to reliably produce a suspension from your tissue sample, no enzymatic dissociation and deactivation necessary – this saves time, effort, money and potential points of error in your workflow.
Publications
Despina Soteriou, Markéta Kubánková, Christine Schweitzer et. al. (2023)
“Rapid single-cell physical phenotyping of mechanically dissociated tissue biopsies”
Nat. Biomed. Eng.
DOI: 10.1038/s41551.023.01015.3
Stefan Scheuermann, Jonas M. Lehmann, Ramkumar Ramani Mohan et. al. (2022)
“TissueGrinder, a novel technology for rapid generation of patient-derived single cell suspensions from solid tumors by mechanical tissue dissociation”
Front. Med., Vol. 9
DOI: 10.3389/fmed.2022.721639
Despina Soteriou, Markéta Kubánková, Christine Schweitzer et. al. (2021)
“Single-cell physical phenotyping of mechanically dissociated tissue biopsies for fast diagnostic assessment”
medRxiv
DOI: 10.1101/2021.11.30.21267075
Stefan Scheuermann, Armin Schäfer, Jens Langejürgen, Christian Reis (2019)
“A step towards enzyme-free tissue dissociation”
Current Directions in Biomedical Engineering, Vol. 5, No. 1
DOI: 10.1515/cdbme.2019.0137
Gallery
Take a look at the new TIGR Tissue Grinder & Dissociator in our new Cell Culture Laboratory in Bremen, the location of our new OLS Life Science School:
Contact
Dr. Kathrin Ringwald
Sales
 Your Partner in Cell Research
Your Partner in Cell Research
Karl-Ferdinand-Braun-Straße 2
28359 Bremen, Germany

OMNI Life Science GmbH
Laufenstraße 90
4053 Basel, Switzerland