3D Cell Culture Bioreactor & Incubator CERO 3D
The CERO 3D Incubator & Bioreactor is a new, revolutionary instrument creating optimal cell culture environment. It offers a special 3D cell culture technology that monitors and controls temperature, pH and carbon dioxide levels. Indeed, this is ideal for stem cells, spheroids, organoids and even tissues.
The CERO 3D Incubator & Bioreactor provides some distinct advantages. First and foremost, it improves viability and maturation with minimized apoptosis and necrosis. The CERO 3D Incubator & Bioreactor has no shear forces, no requirement for embedding substrate, thus providing maximum homogeneity and allowing long-term cultivation for more than one year. Indeed, this will significantly reduce your running costs.
The individually controlled provide highest biomass yields. With volumes up to 50 ml, the CEROtubes, have small fins and a flat bottom allowing mild cultivation conditions in a standardized and reproducible way, with minimum handling requirements.
The CERO 3D Incubator & Bioreactor allows for simplified scale-up and automation platforms, and cost reduction. This is due to its distinct advantages of easy to set-up and simple workflows and minimum hands-on time that can be down to less than 2 minutes per day.
The CERO 3D Incubator & Bioreactor is indeed ideal for a wide variety of applications, from stem cell expansion projects in biobanks, cell-based drug discovery, toxicity testing and regenerative medicine, it is your ideal partner. For stem cells, The CERO 3D Incubator & Bioreactor is able to differentiate in 3 germ layers , provide homogeneous iPSC and ESC 3D aggregates. Your cells can be easily processed directly for differentiation such as for organoids or spheroids as a downstream application in 3D or 2D assay development and avoid technical limitations of long-term culture with high efficiency and standardization.
The CERO 3D Incubator & Bioreactor indeed is an ever-evolving a state-of-the-art dynamic culture system, accelerates your processes reduces costs and hands-on time and allows multiplexing. It provides optimal nutrition, gas diffusion thus increasing size and lifespan of your cultures.
Highlights - 3D Cell Culture Incubator CERO 3D
- Improved viability and maturation
- No embedding substrate required
- Significantly reduced apoptosis & necrosis
- No shear forces
- Long-term cultivation for > 1 year
- Significant reduced running cost
The CERO 3D Incubator & Bioreactor offers a unique 3D cell culture technology to boost your stem cell, spheroid, organoid and tissue research.
Highest levels of homogeneity and viability in long term cultures are just two benefits CERO 3D will provide.
Break the limits in 3D Cell Culture
The CERO 3D Incubator & Bioreactor is a new, revolutionary instrument creating optimal cell culture environment by monitoring and controlling temperature, pH and CO2 levels. 1- 4 individually controlled CEROtubes with a volume of up to 50ml provide highest biomass yields in a standardized and reproducible way, with minimum handling requirements. The CEROtubes, with small fins and a flat bottom, enable gentle cultivation conditions with reduced shear stress to your cells.

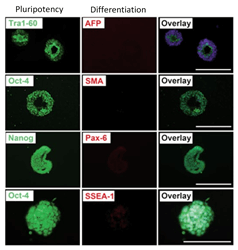
Pluripotent Stem Cells
The CERO 3D Incubator & Bioreactor provides the solution for scale-up and automation platforms, simplification and cost reduction of stem cell expansion projects in biobanks, cell-based drug discovery, toxicity testing and regenerative medicine.
- Microcarrier-free
- Stable pluripotency over many passages
- Easy to set-up and simple workflow
- Free-floating 3D aggregates
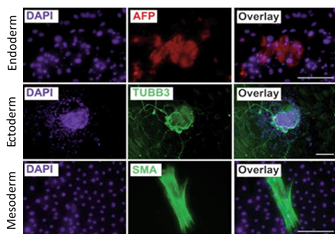
- Able to differentiate in 3 germ layers
- Homogeneous iPSC and ESC aggregates
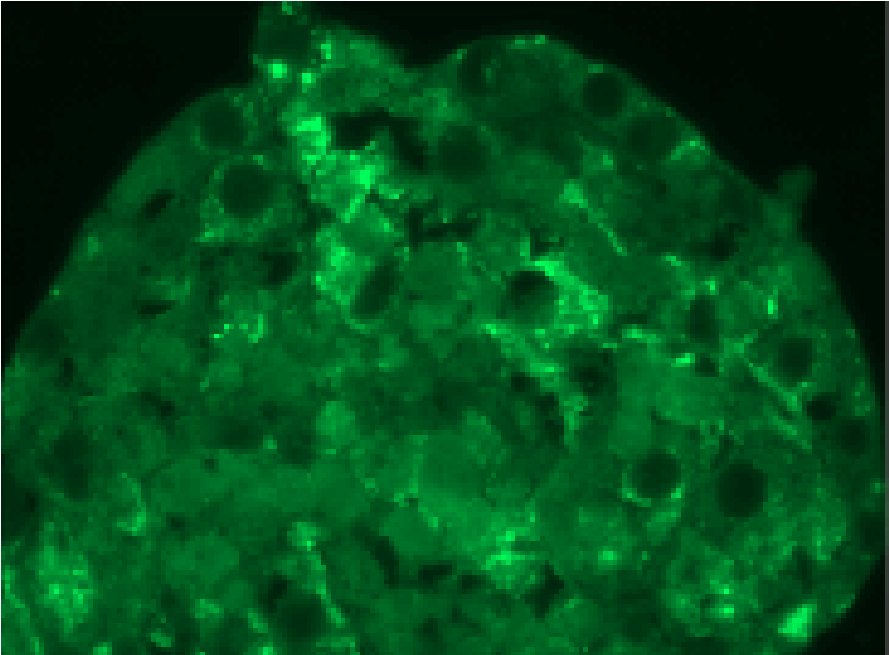
Human iPSC after expansion in CERO 3D
Pluripotent stem cells are directly inoculated as single cells into the CEROtube. During the self-aggregation the cells form homogeneous aggregates which can be expanded over many passages. The biomass increases significantly while only ~2min per day hands on time is required.
The resulting stem cell 3D aggregates can be processed directly for differentiation e.g. organoids or any other downstream application in 3D or 2D assay development.
Pluripotent stem cells expanded in CERO 3D (former name “BioLevitator”) will maintain pluripotency and can be differentiated into all 3 germ layers, as described by Elanzev et. al. 2015; Biotechnol. J. 2015, 10, 1589–1599:
Hepatitis Virus Research Model - Spheroids
The advent of long-term three dimensional cell culture holds a great promise in disease modeling and drug discovery. The cells kept in a 3D environment have the ability to mimic tissue-like structures more efficiently than in traditional 2D monolayer cultures. However, many scientist are struggling with many technical limitations when working with spheroids in long-term cultures.
The CERO 3D Incubator & Bioreactor is a revolutionary technology enabling scientists to perform experiments they were not able to do before.
- No necrosis and apoptosis
- Viability in long-term culture > 80 days
- Improved maturation
- Long-term proliferation
- High homogeneity
- High yield
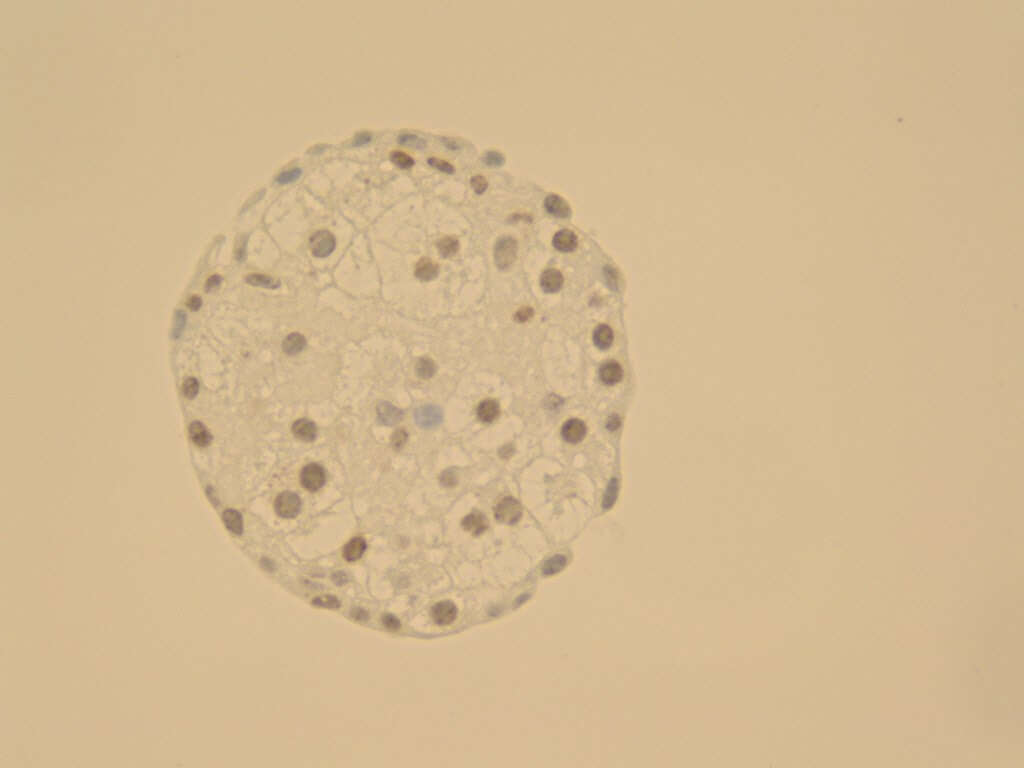
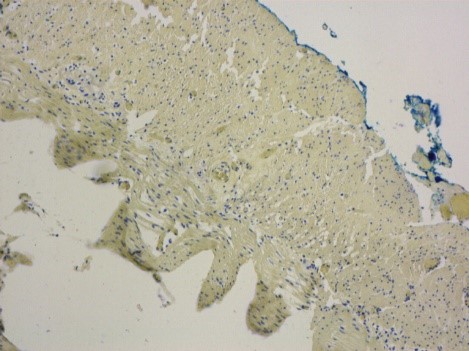
Spheroids from HepG2 cells (hepatocyte cell line) cultivated in CERO 3D for >80 days.
Virus Research
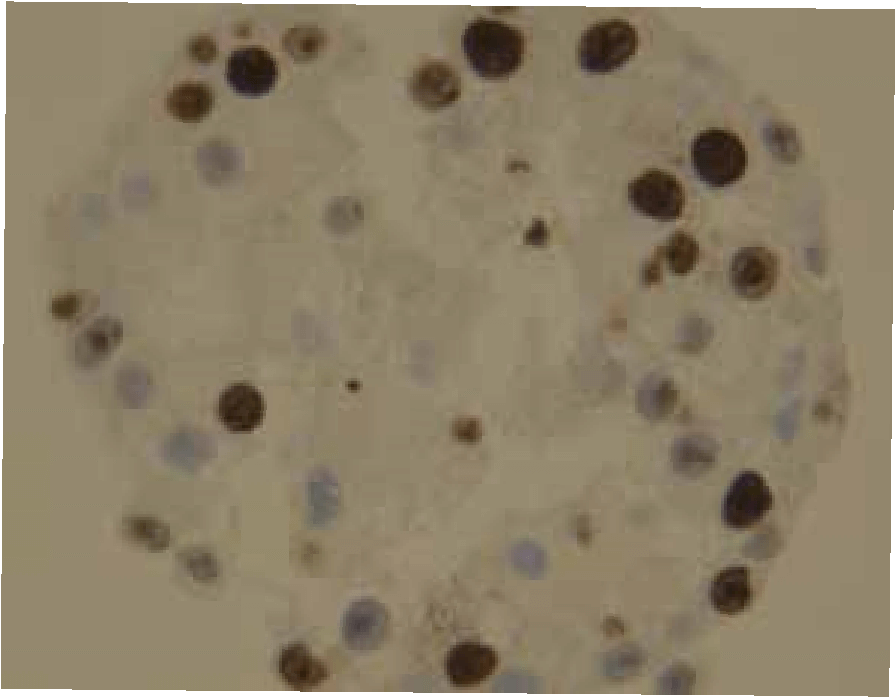
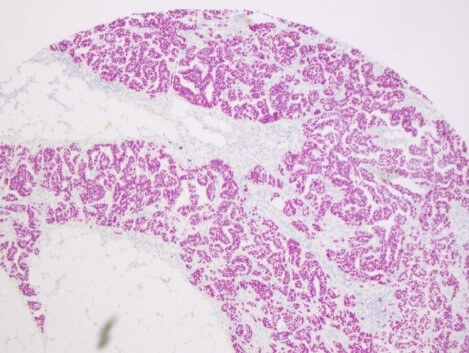
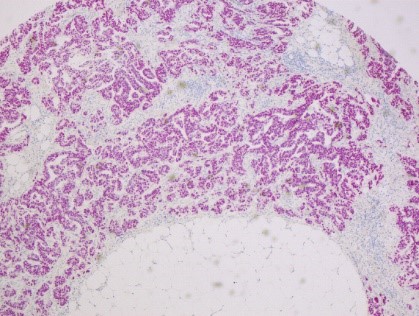
CERO 3D Incubator & Bioreactor enables growth and maturation of spheroids without necrosis and apoptosis while other technologies fail. Therefore, CERO 3D allows to maintain state-of-the-art 3D cultures of Huh7 hepatic cell line (see figure below). The disposable CEROtubes with Hepa filter allow safe virus experiments.

Spheroid from hepatocyte cell line was matured for 20 days prior to exposure to HCV. Infection spreading (rate) was controlled after 24, 48 and 72hours (brown staining)
Cultivating hepatocyte spheroids in CERO considerably improves expansion, differentiation, maturation and hepatic virus infection compared to monolayer culture. Our research takes advantage from healthy cells even from long-term cultures in CERO. Moreover, we are now able to perform 3D long-term culture of human tissue specimen in CERO - a paradigm shift.
Cardiac Tissue Model
Beating Cardiac Bodies – a complete workflow
Stem cell derived cardiomyocytes gain more and more attention in the field of cardiovascular research, disease modeling and drug development.
The CERO 3D Incubator & Bioreactor platform allows the workflow with stem cell expansion in homogeneous aggregates followed by direct induction into a high number of beating cardiac bodies. The expansion of pluripotent stem cells and subsequent cardiac induction/differentiation in CERO 3D results in a much higher cell quality, homogeneity, integrity and yield compared to traditional orbital shakers.
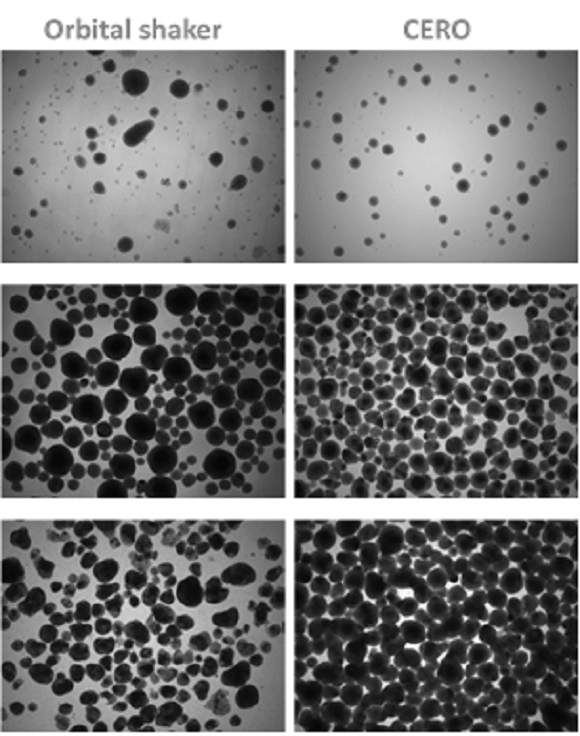
CERO 3D versus Orbital shaker – differentiation of murine embryonic stem cell derived cardiomyocytes 3, 8 and 13 days after cardiac induction.
- Homogeneous 3D aggregates in suspension
- Expansion and differentiation in the same CEROtube
- Easy to handle & standardize
- Highest yield
- No substrate required
- More relevance for drug treatments
- More relevance for disease modeling
Purified and homogeneous cardiac bodies generated in CERO 3D Incubator & Bioreactor are a perfect tool for disease modeling and drug discovery. The pure cardiomyocytes can also be used in 2D downstream monolayer applications.
The overarching aim of all our studies is the identification and development of clinically relevant therapeutic agents for the treatment of human heart disease. To that end we utilize human self-organised cardiac organoids for high throughput drug screening and in vivo genetic models of human congenital disease, as our experimental platform, in combination with adeno-associated virus (AAV) and anti-sense RNAs as therapeutic agents. The CERO 3D has considerably simplified our cardiac organoid generation workflow and enabled us to scale-up organoid production significantly. Organoids generated with CERO 3D reveal improved cellular organisation, and display uniformity and consistency within and across production batches.
Moreover this 3D cultivation system substantially improves viability, maturation and contractility, therefore provides a reliable tool for cardiovascular therapies.

Organoids as Tumor Tissue Model
From Adult Stem Cells to Organoids
The CERO 3D Incubator & Bioreactor is a revolutionary technology for production of organoids from pluripotent or adult stem cells. It offers an efficient, standardizable way to generate and maintain high yields of homogeneous organoids used as a tool in cancer research. Remarkably, the organoids have also a predictive capacity for in vivo response.
- Reduced costs and time
- Increase size & lifespan
- Optimal nutrition & gas diffusion
- Free floating, no shear forces
- High homogeneity and yield
- Multiplexing organoidogenesis
Increase cellular, structural and functional complexity of organoids.
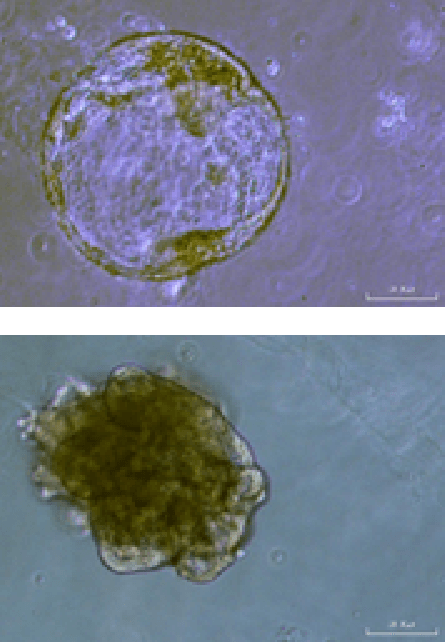
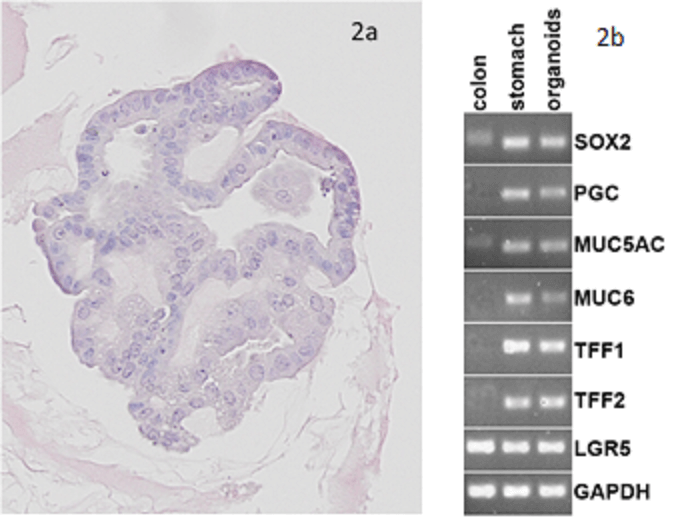
The generation of gastric organoids is a crucial step in the study of Helicobacter pylori infection and gastric carcinogenesis. The CERO 3D Incubator & Bioreactor allows the generation in a much more efficient, reproducible, physiological and cost efficient way compared to any other approach

Personalised and precision medicine
The progress in the development of molecular target therapies has improved the outcome in “in vivo”, however it is because of the tumor heterogeneity in patients that limits the efficiency of those drugs. Thus, to increase the relevance of “in vitro” models, it is particularly important to develop a reliable platform for growing primary tumor cells in a 3D in vitro model, like patient-derived xenografts or organoids. The CERO 3D Incubator & Bioreactor evolving a state-of-the-art dynamic culture system, accelerates precision medicine of primary tumor cells. The CERO 3D offers an efficient and reproducible approach to grow and maintain viable cancer cells in a CERO 3D Incubator & Bioreactor.
Our group is focusing on primary tumor cells grown on 3D polymer scaffolds. For that we are using CERO 3D to develop a model of drug development and precision medicine. It is particularly important that the cells are viable and proliferating in 3D complex structures as seen in tumor tissues. With the CERO 3D we are implementing and developing tools for patient-specific tumor therapy. Furthermore, the cancer cells isolated from fresh tumor biopsies make treatment optimization possible before a therapy is initiated. Here, CERO 3D incubator & bioreactor enables us to develop personalized medicine in a 3D cell culture model.
Publications
Meet the CERO 3D
Latest CERO news

OMNI Life Science Partners with Cell Microsystems
News | Dr. Andreas Friese
Pioneering journey to productive life science advancements
Read more ›Upcoming CERO events
Currently there are no upcoming events. Have a look at our overview of all events.
Downloads
Application Note
Generation of HepaRG Spheroids in the CERO
Scientific Poster: "Establishment and validation of the 3D cultivation system“CERO” for hepatic spheroids to study viral hepatitis"
Scientific Poster: "Establishment and validation of the 3D cultivation system“CERO” for hepatic spheroids to study viral hepatitis", presented at the 2018 EMBL Symposium "Organoids: Modelling Organ Development and Disease in 3D Culture"
Protocol
Expansion Protocol Pluripotent Stem Cells (iPSC and ESC)
Get in touch
Amir Keric
Sales
 Your Partner in Cell Research
Your Partner in Cell Research
Karl-Ferdinand-Braun-Straße 2
28359 Bremen, Germany

OMNI Life Science GmbH
Laufenstraße 90
4053 Basel, Switzerland