Cell culture is a crucial step for all stages and applications of life science research. Whether you are creating a biobank, running diagnostics, studying complex immune-oncology of patients or running drug discovery processes, quality of cell culture has to be at its best.
Improper handling, maintenance and most importantly monitoring of cell culture concentrations can lead to unhealthy cells that alter results and produce artifacts.
Allowing cells to overgrow before an experiment can produce cells that are about to die or undergo apoptosis. Over a period of time, one might end up selecting cells that are more fit to handle harsh environmental conditions, with a migration and morphology profiles that are different from healthy cells at physiological conditions.
For basic cell culture, monitoring cells through the process of live cell analysis is an ideal way to go. Our Live Cell Analysis System xCELLigence, is your ideal cell culture partner for every step of the way. Using the xCELLigence Real-Time Cell Analyzer, you can monitor your cell numbers, cell-cell contacts, cell morphology over time as well as their migratory and invasive properties.
For virology research, virus-infected cells often display the cytopathic effect (CPE), reflecting morphological changes, cell fusion, lysis or the formation of inclusion bodies. Our Live Cell Analysis System xCELLigence allows detection of subtle changes occurring during CPE, like changes in cell-surface adhesion that can easily be missed by microscopy.
Indeed, the xCELLigence Real-Time Cell Analyzer allows you to study cells in co-culture as well. Secretory proteins such as cytokines and chemokines, can lead to induction or reduction of cell proliferation, altering in normal cell development and tumorigenesis. This can be helpful in studying immune-tumor interaction in immune-oncology as well.
Highlights - Live Cell Analysis System xCELLigence
- Continuously monitor live cell proliferation, morphology and viability
- No cell labeling required
- Impedance-based real-time analysis
- Different throughputs from 16 to 4×384 well formats
- Kinetic read-out – capture both short (sec) and long-term effects (days)
- Easy – Walk-away operation
- Exceeds limitations from endpoint-assays
Live Cell Analysis with xCELLigence®
Live cell analysis with xCELLigence®– much more than an endpoint assay can deliver.
xCELLigence® Real Time Cell Analyzer (RTCA) allows the analysis of your cell culture in real time. A huge variety of cell-based assays, such as proliferation, adhesion, cytotoxicity, migration, morphological changes and more can be performed (for detailed information please visit our download section). Furthermore, xCELLigence® is scalable. Depending on your throughput, evaluate 16 up to 4 x 384 well-plates simultaneously.
Highly Sensitive: The impedance-based technology is highly sensitive, allowing very low effector to target ratios. Continuous cell monitoring in real time enables investigation of short- and long-term effects. As a result, a full kinetic read out of the cellular behaviour is obtained- objective and quantitative.
Non-Invasive: xCELLigence® requires no cell labeling, which increases the physiological relevance and reduces the workload.
Easy to operate: Now run your cell-based assays with xCELLigence® RTCA easily. Literally! Click on RUN button and RELAX
How does it work?
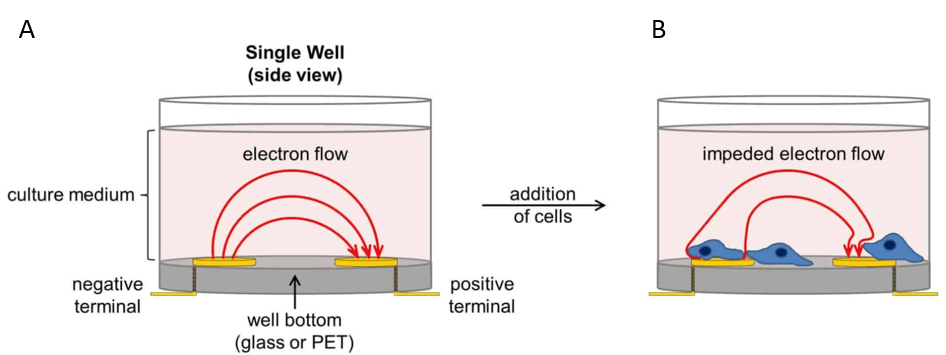
xCELLigence® technology is based on impedance sensing. The E-Plates® form the heart for this technology. The functional unit of the E-Plates® are the gold microelectrodes, which cover 70 – 80 % of its well-bottom area (Figure 1).

For impedance measurement, a very low electric current is applied to the plates, flowing through an electrically conductive solution (cell culture media) from one microelectrode to the other (Figure 2A). As soon as cells adhere to these microelectrodes they act as electrical insulators, thereby influencing the impedance signal (Figure 2B). Importantly, cell behaviour is not affected by the gold microelectrodes or the electrical current which is applied.
How does the analysis look like?
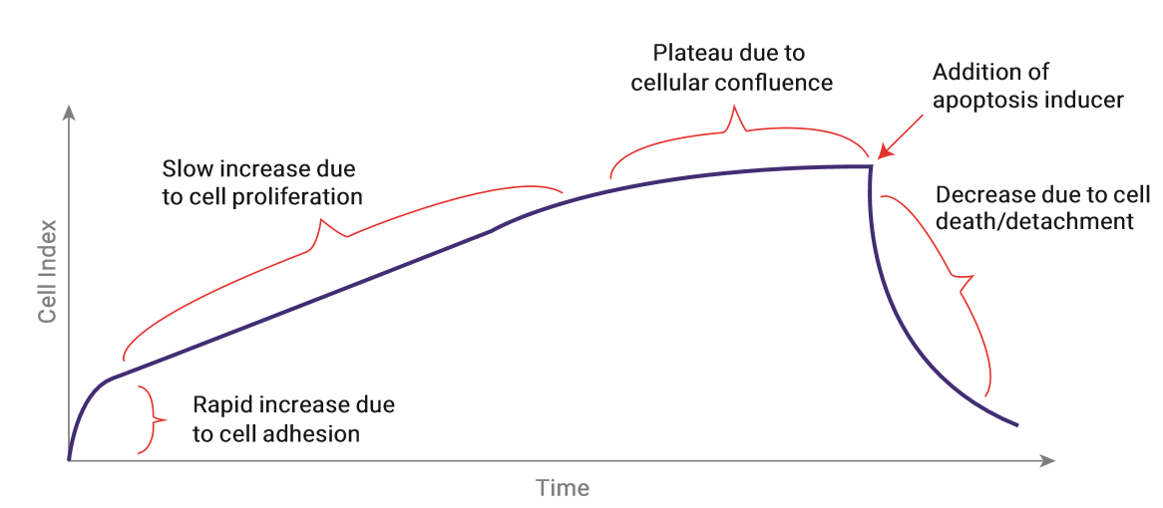
The change in impedance is reported as a dimensionless parameter called Cell Index (CI, CI= impedance at time point n – impedance without cells/nominal impedance value).
Figure 3 shows a typical course of the CI value during an apoptosis assay. Directly after cell seeding, cells start to adhere, thereby inducing a rapid increase of the CI value. The adhesion phase is followed by a phase where cells grow exponentially, indicated by a continuous increase of the CI value. When cells are confluent the CI value cannot further increase, thus forming a plateau. Upon the induction of apoptosis by a certain treatment cells start to die and subsequently detach from the well bottom. The reduction of adherend cells is reflected by a decrease in the CI value.
Investigate a huge variety of cell-based questions
xCELLigence® RTCA technology allows detection of minimal cellular changes, involving strength of adhesion, cell number, cell morphology and cell-cell-contacts. All these properties influence cellular impedance signal that is continuously measured as Cell Index. Thus, xCELLigence RTCA provides deep insight into whole kinetics of cellular behaviour without any cell-labelling and with only minimal hands-on-time.
Migration and Invasion

Contact us directly to learn more about the RTCA DP.
Quantitative. Reproducible. Label-free.
Exeed limitations of normal boyden chamber assays and track cell invasion and migration in real time.
Migratory and invasive properties of cells can easily be analyzed using RTCA CIM (Cell Invasion & Migration) plates. CIM plates are composed of an upper chamber, containing cells of interest and a lower chamber containing certain cell attractants. These chambers are separated by a microporous membrane (pore size about 8 µm).
Similar to a boyden chamber assay, cells actively pass the membrane towards a chemoattractant in the lower chamber. Cells passing through the pores of the membrane are immedeatly detected by gold electrodes, covering the lower side of the membrane. These gold electrodes measure the change of impedance, which increases with the number of migrated cells attaching on the gold microelectrodes on the lower side of the membrane.
No need for time-consuming manual detachment, staining and counting of cells from lower sides of membranes.

- Transverse section of a RTCA CIM plate
- Transverse section of a single well of a CIM plate: a) Upper compartment with cells of interest; b) Microporous membrane with gold microelectrodes underside; c) Lower compartment containing chemoattractant
- Cell, migrating through a pore of the CIM plate membrane
- Lower side of CIM plate membrane, covered with gold microelectrodes and having first cells attached that migrated through the membrane towards the chemoattractant in the lower well.
- Transverse section of a single well of a CIM plate with adherent cells in the upper well and cells that already migrated to the lower well and attach to gold microelectrodes, covering the lower side of the microporous membrane.
To investigate cell invasion processes, CIM plates can easily be coated with different matrices, like matrigel, collagen etc.
Further analysis of the migrated/invaded cells or the correlation of the impedance signal with direct cell numbers can easily be realized by simply staining the cells on the membrane and having a closer look under a microscope.
Virology Research
Analyze virus-induced effects in an easy and more efficiant way.
xCELLigence technology is the perfect tool to address typical questions in the field of viral research:
- Determination of virus infection and virus titer
- Detection and quantification of neutralizing antibodies
- Antiviral drug screening
- Testing virucide efficacy
- Characterizing virus quality and fitness
- Oncolytic viruses
Virus-infected cells often display a so called cytopathic effect (CPE), reflecting morphological changes, cell fusion, lysis or the formation of inclusion bodies etc.
The extraordinary sensitivity of RTCA xCELLigence technology enables not only the detection of cell rounding or lysis but even allows detection of subtle changes occurring during CPE, like changes in cell-surface adhesion that can easily be missed by microscopy.
Continues measurement of cellular impedance facilitates tracking of viral CPE in unpredicted detail and provides quantitative kinetics that can be used for various applications.
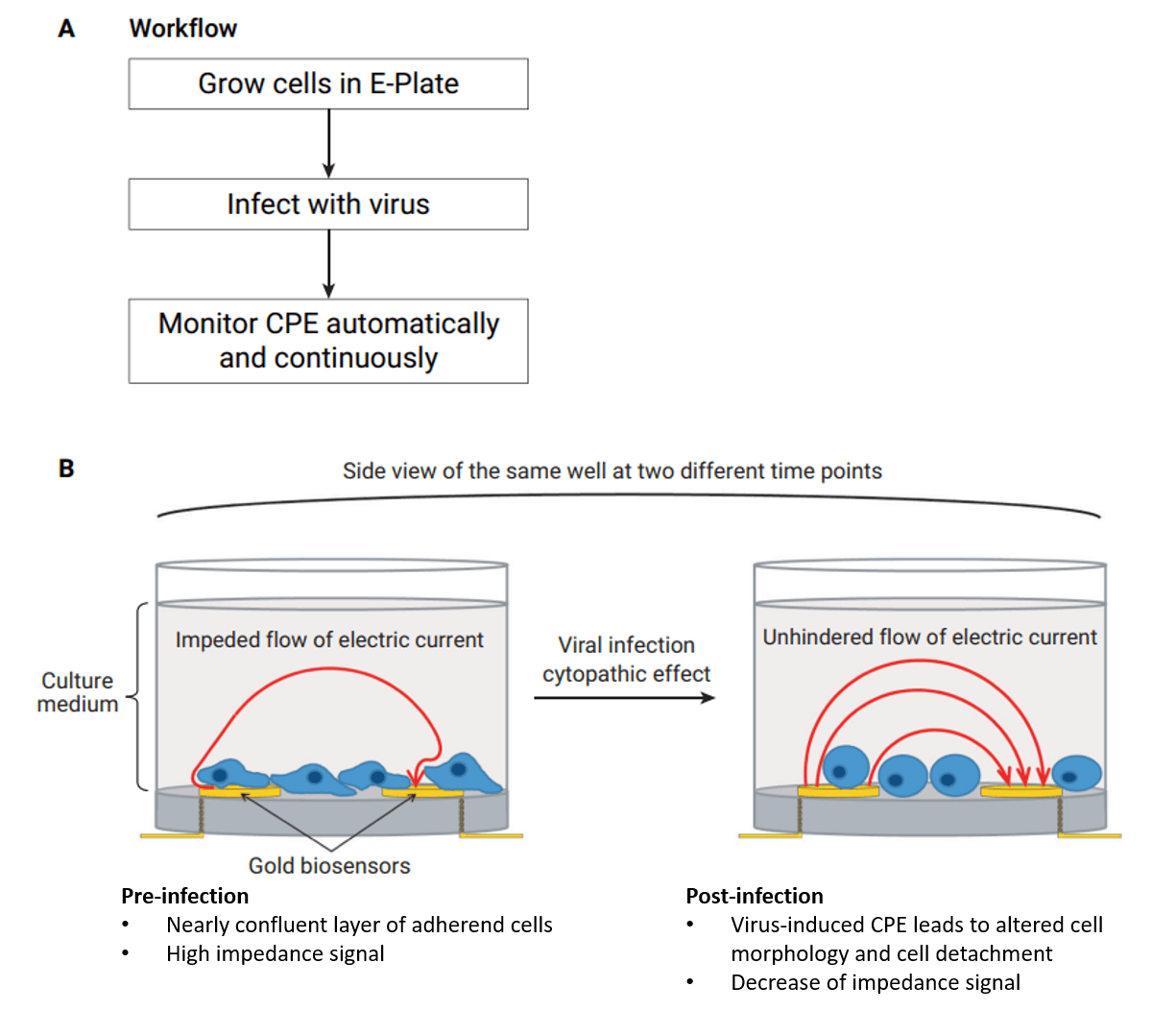
Easy handling and automated data collection
The picture shows a schematic representation of a typical experimental workflow to monitor viral CPE in real time using RCTA xCELLigence system. A) Easy workflow: Cells are grown in E-plates and infected with viruses. Automated and continuous reading of CPE-induced changes of electrical impedance. B) Virus-induced CPE, indicated by changes in cellular morphology, cell surface attachment, cell-cell adhesion and decreased number of living cells, lead to a decrease of electrical impedance, measured by gold biosensors in real time.
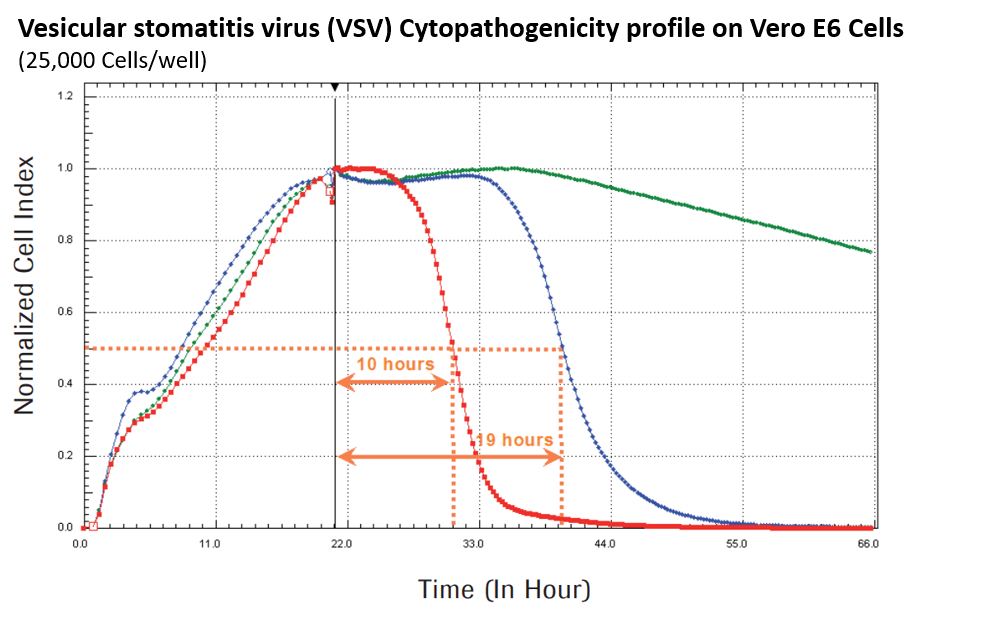
Data from App. Note 9: A New Way to Monitor Virus-Mediated Cytopathogenicity
Dynamic monitoring of Vero E6 cells during VSV infection.
Vero E6 cells were seeded at a density of 25.000 cells/well and analyzed every 15 minutes with a RTCA xCELLigence SP system. During the first 20 hours after cell seeding the Normalized Cell Index (NCI) values were increasing due to increased cell adhesion and exponential cell growth. Cell Index values reflect changes in cellular impedance depending on strength of adhesion, morphology, cell number etc. When cells reached confluence, the virus was added as indicated by the black vertical line. The virus-induced CPE leads to cell detachment and is nicely represented by a decrease of NCI values. Interestingly, the time course of the CPE clearly correlates with the virus titer (green: control (no viral infection); blue: 80,000 PFU VSV; red: 800,000 PFU VSV). Additionally, RTCA software offers a huge variety of analysis tools. Here, automated calculation of CI50 values represent the time point when the Cell Index had decreased to 50 % of the maximum Cell Index, indicated by the dotted orange lines.
Investigate Cytotoxicity in Real Time
Get the most out of it.
RTCA xCELLigence technology provides a great tool to unravel the entire course of a cytotoxic response without losing any meaningful time points. Profit from easy handling and extraordinary sensitivity. Automated and continuous measurements ensure highly reproducible and quantitative data.
No need for cell labelling or counting.
xCELLigence Technology is based on impedance measurement. Microelectrodes, integrated into the bottom of each well of the tissue culture E-Plates, detect minor changes of cellular impedance that occur upon changes in cell number, cell morphology, cell surface attachment, etc.
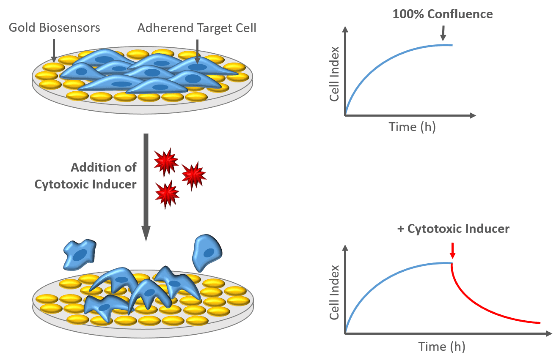
Cytotoxicity
In general, cytotoxic responses lead to an altered cellular morphology, often accompanied with cell detachment from the well bottom. Cells that loose their contact to the gold biosensors on the plate buttom are no longer detected as electrical insulators, resulting in a decrease of Cell Index values.
Analyze various cytotoxic inducers
Cytotoxicity can be caused by different inducers, like chemical substances (e.g. drugs), immune cells (cytotoxic T-cells, natural killer cells), viruses or irradiation. Unseen or novel effects of all these cytotoxic inducers can easily be explored using xCELLigence systems.
- Compound-induced Cytotoxicity
Compound-induced cytotoxicity of Hela cells
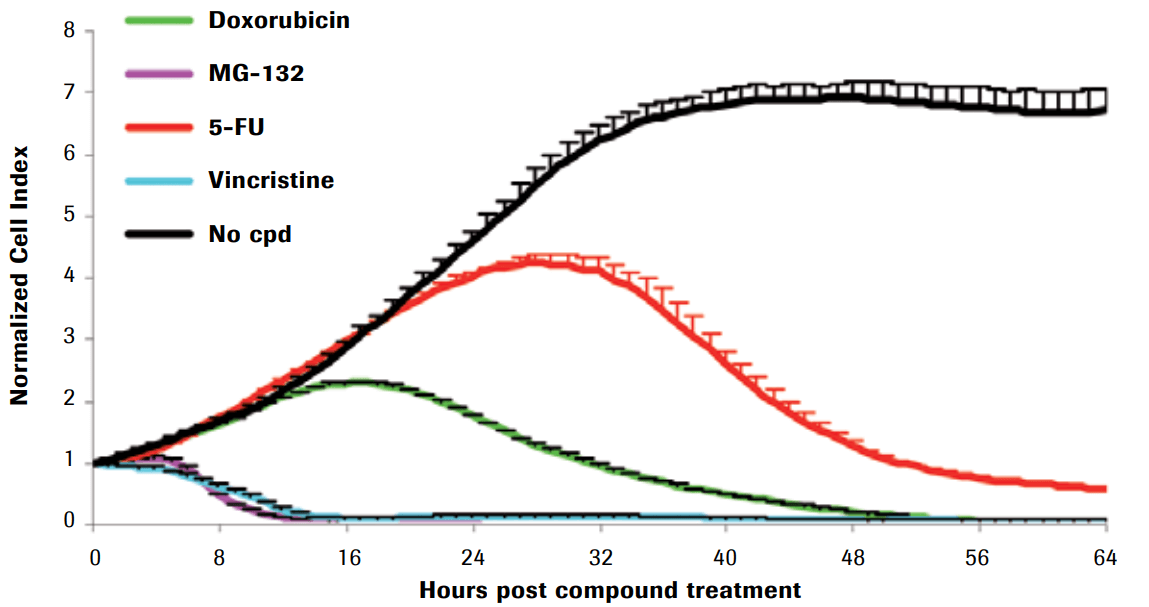
Compound-induced cytotoxicity of Hela cells
Here, Hela cells were seeded in an E-Plate 96 for 24 hours and subsequently treated with different cytotoxic compounds. Cell viability was monitored every 15 minutes. While Cell Index values of non-treated control cells increased due to unhindered cell proliferation (black curve), decreasing Cell Index values of treated cells indicated cell death, morphological changes and cell detachment. Notably, even though all compounds were cytotoxic agents, the kinetic profiles provided by xCELLigence system revealed highly different rates and dynamics of cytotoxicity. This shows diverse modes of cytotxic actions, like DNA damage (doxorubicin, 5-FU), proteasome inhibition (MG-132), anti-mitosis (vincristine).
- Cell-Mediated Cytotoxicity
Easy as it can be: To investigate cell-mediated cytotoxicity, xCELLigence technology benefits from different adhesion properties of target and effector cells. While adherent target cells induce high Cell Index values, non-adherent effector cells, like natural killer (NK) cells, remain in suspension and hardly interact with the microelectrodes. These non-adherend effector cells will lead to no or only minimal changes of Cell Index values. Remarkably, upon addition of non-adherent effector cells to a confluent layer of target cells, Cell Index values will decline due to the cytolytic activity of the effector cells. Decreasing Cell Index values indicate changes in number, size and shape of adherent target cells and are dependent on the amount and specificity of the added effector cells.
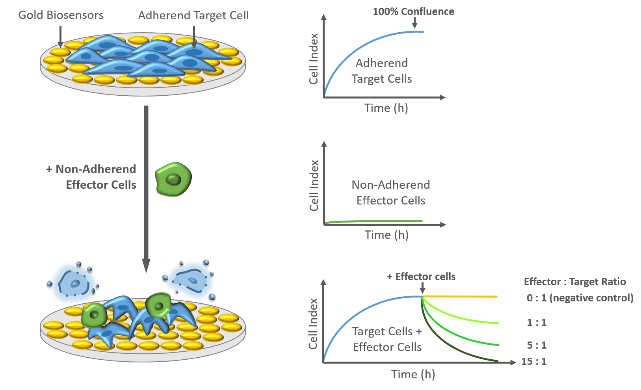
Cell-mediated Cytotoxicity
Continues measurement using xCELLigence System uncovers the kinetics of cell-mediated cytolysis in a label-free and quantitative manner with minimal hands-on time. Highly sensitive gold biosensors enable detection of effects from very low effector to target (E:T) cell ratios.
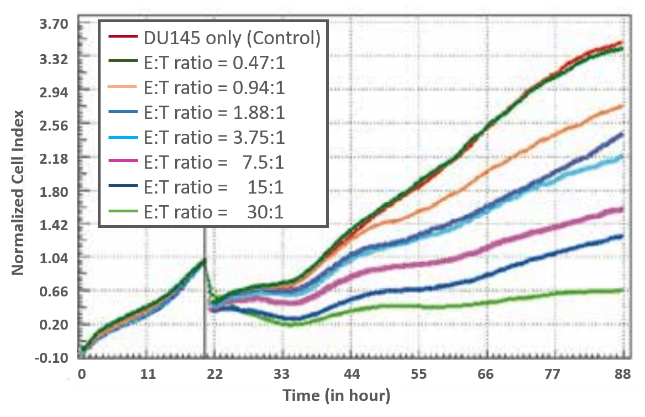
ADCC of prostate cells induced by NK 92 cells
To analyze cell-mediated cytolytic activity, the prostate cancer cell line DU145 was used as target cells, seeded into an E-Plate 96 and monitored every 15 minutes using xCELLigence MP system. Cell Index values increased upon cell attachment, followed by cell proliferation. When target cells reached their logarithmic growth phase, NK92 effector cells stably overexpressing FcγR III (CD16) were added as indicated by the black vertical line at different effector-to-target (E:T) cell ratios.
Cell Index values were normalized to the time point of effector cell addition. Directly after addition of NK92 cells, the Cell Index values slightly decreased due to temperature changes within the medium. Interestingly, after one hour the decline of Cell Index values highly correlated with the increase in the number of effector cells.
RTCA MP (Multi-Plate)Learn more or contact us directly for information on the RTCA MP.
We are investigating the activity of Natural Killer (NK) cells. When using adherent target cells, the xCELLigence system provides label free and real time data about the NK cell killing activity. Especially when combining NK cells and cytotoxic substances we can determine the individual contribution of each factor, but also synergistic effects on target cell killing. Additionally, we are using the xCELLigence system to determine NK cell adhesion and activation in a time-resolved fashion.

- Antibody-dependent cellular cytotoxicity (ADCC)
Antibody-dependent cellular cytotoxicity is a mechanism of cell-mediated immune defense that involves active lysis of target cells, mediated by effector cells from the immune system, like natural killer (NK) cells. Specific antibodies, presented by the target cells are recognized by antigens presented on the effector cells. Once the NK cell bounds to the target cell, cytotoxic factors are released inducing target cell death. Cytolytic activity is represented by decreasing Cell Index values and is highly correlated with the amount of added effector cells and their specificity towards the antibody presented by the target cells.
Hence, xCELLigence RTCA is a suitable tool to evaluate complete kinetics of ADCC throughout the entire course of an experiment.
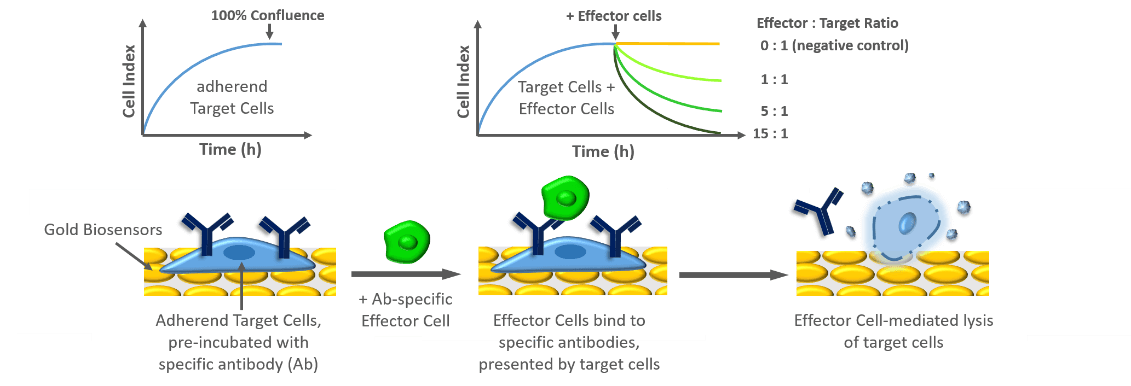
Schematic representation of analyzing ADCC using xCELLigence Technology
The picture below show example data for ADCC of DU145, induced by NK92 cells:
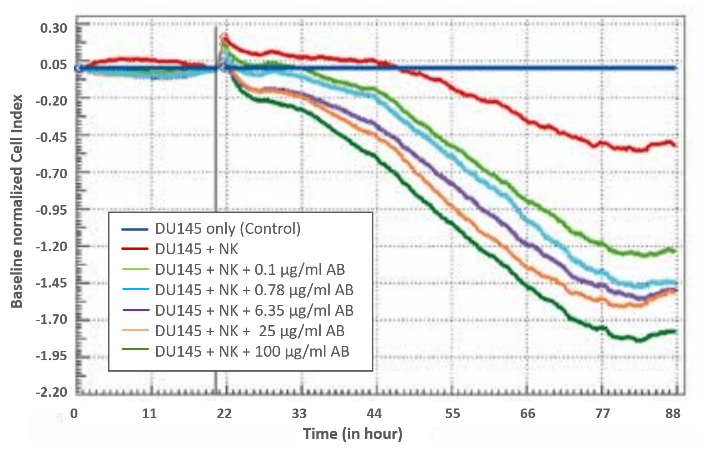
ADCC of prostate cells induced by NK 92 cells
In this example data, ADCC of NK92 cells for the prostate cell line DU145 as target was investigated. These target cells were pre-incubated with different amounts of anti-IGF-1R monoclonal antibody that binds to the extracellular human IGF-1R and the Fcγ receptor III (FcγR III) expressed on NK92 cells.
5.000 DU145 cells were seeded into each well of an E-Plate 96 and monitored every 15 minutes using a xCELLigence MP system. Target cell adhesion and proliferation resulted in increasing Cell Index values. In the diagram, Cell Index values of non-treated target cells (dark blue curve) were set as baseline. Additionally, Cell Index values were normalized to the time point of effector cell addition (20.5 h after cell seeding). Indeed, addition of NK cells (E:T ratio of 1.88:1) resulted in a decrease of Cell Index values indicating cell detachment due to the cytolytic activity of NK cells. Nevertheless, pre-incubation of the target cells with anti-IGF-1R led to enhanced cytolytic activiy, which strongly correlated with the amount of anti-IGF-1R.
- Irradiation-induced cytotoxicity
Irradiation-induced cytotoxicity of HEK-293 cells:
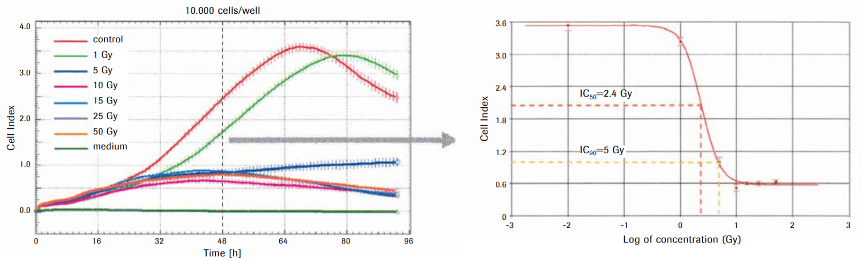
Data from Application Note „Irradiation-Induced Cytotoxicity“
To investigate irradiation-induced cell damage, HEK-293 cells were plated into a E-Plate 96 and treated with different doses of γ-irradiation (1 – 50 Gy). Cell viability was continuously analyzed every 15 minutes using xCELLigence SP system. Irradiation induced a dose-dependent decrease of the Cell Index values of the irradiated HEK-293 cells, reflecting the severity of cell damage. Low doses of irradiation resulted only in a slight decrease and a short delay before Cell Index values of untreated control cells (red curve) were reached again. This might be explained by irradiation-induced DNA damage that could be repealed by cellular DNA repair mechanisms. In contrast, irradiation of five gray units (Gy) and more completely stopped cellular proliferation, suggesting irradiation-induced DNA damage to a level that leads to irreversible cell senescence (5 Gy) and massive cell death (10 Gy and more). Toxicity was quantified by calculating the inhibitory concentration (IC) from the Cell Index values of the dose-response curve 48 hours after irradiation.
Indirect Co-Culture
Cells are able to influence each other – even without direct interaction. Therefore, they communicate via small secreted signalling proteins, such as cytokines and chemokines. These soluble factors can modulate the fate of target cells in different ways, like induction or reduction of cell proliferation, which plays a central role in normal cell development and tumorigenesis.
To uncover the impact of soluble signalling proteins, xCELLigence offers a special device, called E-Plate Insert, that is inserted into a normal E-plate and allows indirect co-culture of target and effector cells. A microporous membrane ensures communication via soluble signalling proteins but prevents cells from passing to the other side.
Hence, the effect of signalling proteins, released by indirectly co-cultured effector cells, can easily be monitored in real time using xCELLigence RTCA.


Indirect co-culture of breast cancer cells with hormon-producing effector cells:
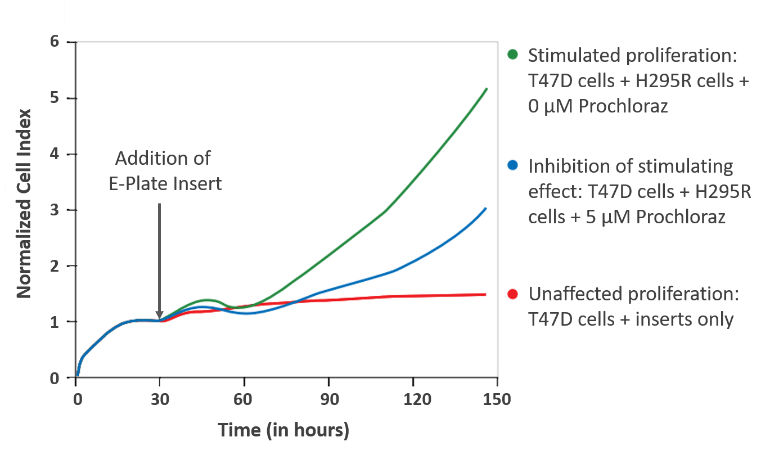
Adapted from „Co-culture Device Protocol“
To investigate even more complex interactions of cells, growing in three-dimensional spheroids or organoids, the CERO would be the ideal device.
In this example data, the breast cancer cell line T47D was used as target cell and indirectly co-cultured with steroid-producing adrenocortical effector cells H295R. Real-time monitoring using xCELLigence RTCA revealed increased proliferation of T47D cells upon indirect co-culture with H295R cells. T47D cell proliferation is represented by increasing Cell Index values (green curve). This effect could be highly reduced by addition of hormone synthesis inhibitor Prochloraz (blue curve). As control, T47D cells were incubated with only the E-Plate Insert, which had no impact on cell proliferation (red curve) as shown by stable Cell Index values.
General overview of applications
Application Book
Application Book
Cancer Immunotherapy
Handbook
Cancer Immunotherapy
Brochure
Liquid Tumor Immunotherapy
Application Note
Evaluating Functional Potency of Immunotherapies Targeting Tumors of B-Cell-Origin
White Paper
Multifunctional Potency Assays for Cancer Immunotherapy
Brochure
Immunotherapy Software
Infectious Diseases
Handbook
Infectious Diseases
Bacterial Biofilms
Application Note
Bacterial Biofilms
xCELLigence RTCA Family
Depending on your throughput different xCELLigence® RTCA systems are available.
RTCA S16
With one single 16-well plate the S16 is the smallest member of the xCELLigence® RTCA family. It is the pefect device for assay development and optimization.

RTCA DP (Cell Invasion and Migration)
Highest flexibility: Three 16-well plates can be operated seperatly or together, giving you the possibility to run 48 wells in parallel to maximize productivity.
Migration and Invasion: Besides the „normal“ E-Plates®, the DP system can be operated with CIM (Cell Invasion and Migration) Plates® to investigate migration and invasion of cells.
Download RTCA DP Brochure

RTCA SP (Single Plate)
In the SP (Single Plate) model one 96-well plate can be analyzed. In approximately only 7 seconds the whole 96 well plate is measured.
Download RTCA SP Brochure

RTCA MP (Multi-Plate)
The MP system offers the opportunity to analyse from one up to six 96-well plates. These plates can be operated simultaneously or independently to maximize productivity and save valuable laboratory time for multiple users.
Download RTCA MP Brochure
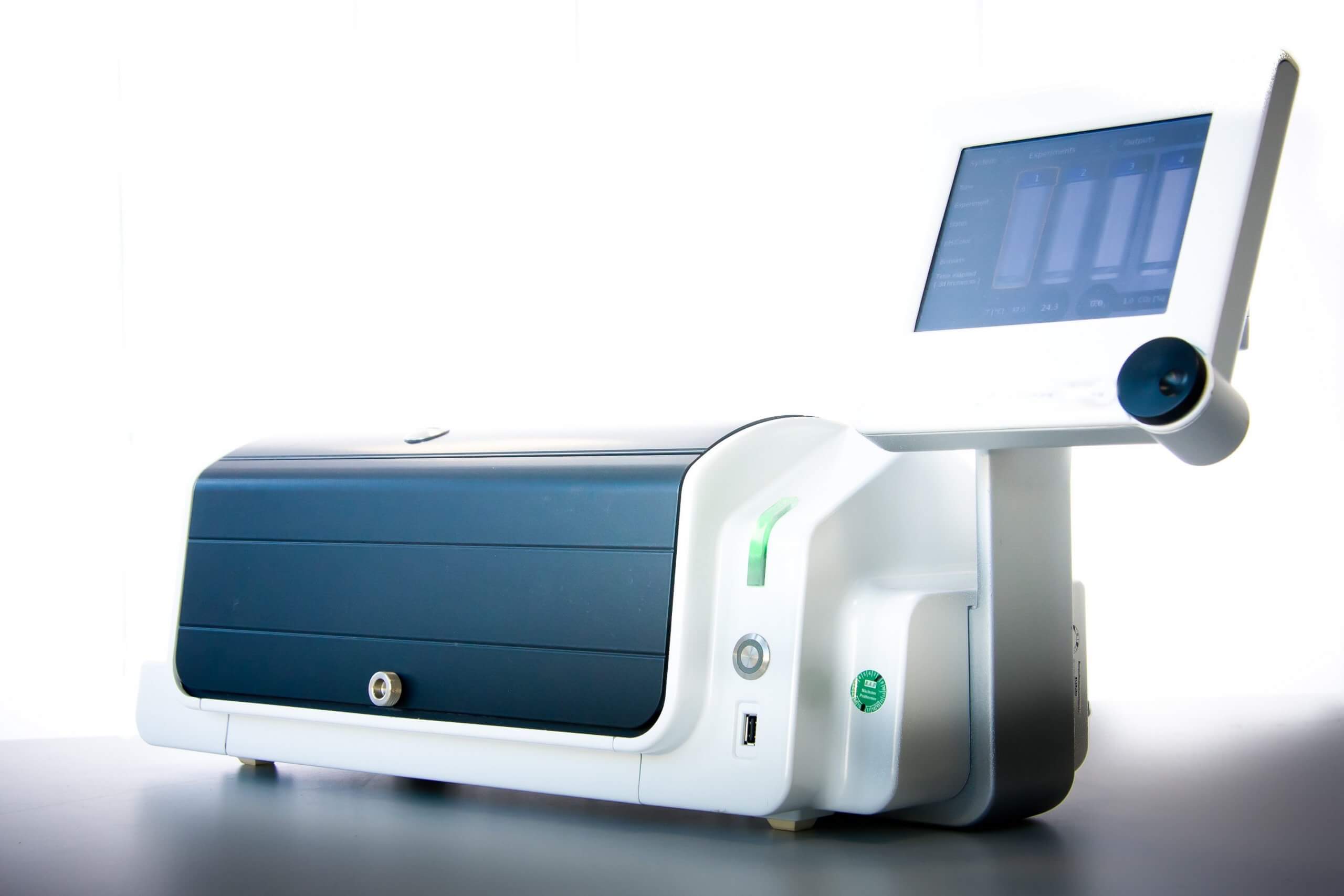
RTCA HT (High Throughput)
The HT system allows high troughput analysis of a 384-well plate. To maximize the throughput, up to four instruments can be integrated and monitored by one control unit. This gives you a maximal screening capacity of 1536 wells in total.
Download RTCA HT Brochure
RTCA eSight (Imaging & Impedance)

Visit our product site and learn more about the RTCA eSight.
The eSight is the first system of its kind that combines high sensitivity of xCELLigence impedance-based measurement with the benefit of live cell imaging. These two modes can be evaluated simultaneously, providing an extraordinary insight in cellular behaviour and underlying mechanisms. Analyze up to five 96-well plates in parallel or individually – in real time, using bright field or up to three colours (green, blue, red).

RTCA Cardio (Cardiomyocyte Contractility)
Analyze cardiomyocyte contractility and viability in real time. Similar to the other RTCA xCELLigence® members, the Cardio system is based on noninvasive measurement of electrical imedance to monitor changes in cellular morphology, proliferation and attachment.
The difference to the other RTCA systems is a higher acquisition rate of 12.9 milliseconds. This high acquisition rate in the range of milliseconds enables the detection of morphological changes that are due to cardiomyocyte contractility. The cycle of cardiomyocyte contraction and relaxion is recorded as a rhythmic chance in cellular impedance.
Furthermore, the Cardio model allows investigation of cell viability. This facilitates short- (second) and long- (days) term investigation of cardiotoxicity which becomes highly relevant for clinical safety testing.
The Cardio system can be used to study stem cell-derived, induced pluripotent stem cell (iPSC)-derived, or primary cardiomyocytes.
Cells are analyzed in a 96-well format.
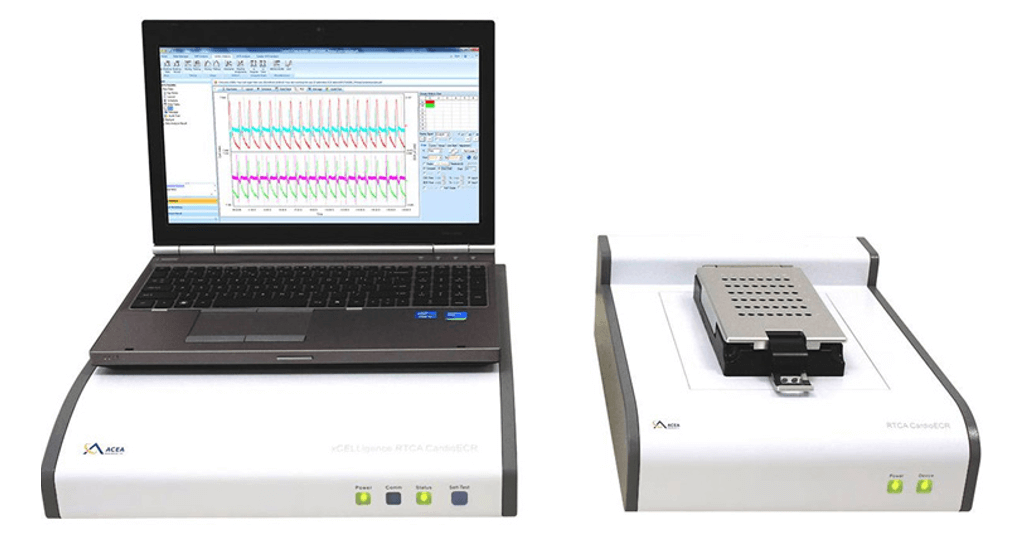
RTCA Cardio ECR (Cardiomyocate Contractility and Electrical Activity)
Analyze cardiomyocyte contractility, viability and electrophysiology in one run.
Similar to the Cardio model, the Cardio ECR system has a second measurement mode within the millisecond range that enables the detection of cardiomyocyte contractility.
As additional benefit, the Cardio ECR (ExtraCellularRecording) model is equipped with an enhanced acquisition rate of 1 millisecond. Furthermore, it is able to measure the extracellular field potential at 10 kHz and to perform cardiomyocyte pacing. This pacing function supports the development of functionally mature cardiomyocytes.
These extra functiosn of the Cardio ECR model provide an unpredicted deep insight into cardiomyocyte health, cellular responses and underlying mechanisms to certain drugs. Therefore, xCELLigence Cardio ERC system is of relevance for any kind of in vitro assays, investigating drug induced cardic liability.

RTCA ePacer (Cardiomyocyte pacing)
The ePacer is the system of choice to produce functionally mature hiPSC cardiomyocytes in an easy and efficiant way.
Less cell stress: Precise and standardized pacing conditions result in improved maturation of hiPSC cardiomyocytes in only 2-3 weeks. From just a few wells up to six 48- or 96- well E-Plates® you can analyze cardiomyocyte beating pattern.
Integrate the ePacer into your existing workflow: Combine the ePacer with your existing readouts, such as calcium measurements, microscopy, voltage sensitive dyes or plate readers.
Furthermore, investigate contractile and electrical responses of long-term paced hiPSC cardiomyocytes with the xCELLigence RTCA CardioECR system. Contractile responses only can be explored using the xCELLigence RTCA Cardio system.
AccuWound® 96
Investigate Cell Migration and Invasion in an incredible easy and highly reproducible manner.
AccuWound®96 automatically produces 96 identical scratches in a 96 microtiter plate. Unlike in traditional scratch assays / wound healing assays, these scratches are highly precise with a coefficent of variation of around 4 % compared to 20% which is typical for traditional scratch assays.
In combination with xCELLigence RTCA systems, cell migration/ invasion can be investigated in real time. Giving you precise and objective insight into the full kinetic of scratch closure.
We are studying oncogenic and metastatic signaling pathways in pancreatic carcinoma cells. Cell migration assays using the xCELLigence DP system have proven indispensable for our work, as it allows us to generate valuable, easily quantifiable kinetic data, combined with a simple and time-saving setup procedure.
Latest xCELLigence news

New OLS Brochure Available
News | Fabian Stautz
OLS released the new 2023 brochure with a comprehensive overview over the key focuses and all available products.
Read more ›
Real-Time Cell Analysis and Monitoring of Virus-Mediated Cytopathic Effects
Live Cell Analysis | Dr. Kathrin Ringwald
In the current times, where several of us are fighting the race against the time to find the cure and prevention for COVID-19, the evaluation and the need for better technologies for virus-mediated cytopathogenicity have become more and more of relevance. The xCELLigence real-time cell analysis (RTCA) from Agilent technologies proves to be one such technology, making use of non-invasive electrical current based measurements.
Read more ›Upcoming xCELLigence events
Currently there are no upcoming events. Have a look at our overview of all events.
Devices
| Name | Article Number | Description | Manufacturer's number | Quote |
|---|---|---|---|---|
| xCELLigence MP | 2801003 | xCELLigence RTCA MP, Real Time Cell Analyzer | ACEA # 00380601040 | Quote |
| xCELLigence SP | 2801000 | xCELLigence RTCA SP, Real Time Cell Analyzer | ACEA # 00380601030 | Quote |
| xCELLigence HT | 2801012 | xCELLigence RTCA HT, Real Time Cell Analyzer | ACEA # 00380601070 | Quote |
| xCELLigence DP | 2801006 | xCELLigence RTCA DP, Real Time Cell Analyzer | ACEA # 00380601050 | Quote |
| xCELLigence S16 | 2801919 | xCELLigence RTCA S16, Real Time Cell Analyzer | ACEA # 00380601310 | Quote |
| xCELLigence cardio | 2801009 | xCELLigence RTCA Cardio, Real Time Cell Analyzer | ACEA # 00380601060 | Quote |
| xCELLigence cardio ECR | 2801190 | xCELLigence RTCA Cardio ECR, Real Time Cell Analyzer | ACEA # 00380601210 | Quote |
| xCELLigence ePacer | 2801351 | xCELLigence RTCA ePacer, Real Time Cell Analyzer | ACEA # 00380601630 | Quote |
Consumables
E-Plate 16 for the xCELLigence DP
Price on request
E-Plate 16 for the xCELLigence DP 1×6 Plates
E-Plate 96 for the xCELLigence SP/MP
Price on request
E-Plate 96 for the xCELLigence SP/MP 1×6 Plates
CIM Plate 16 for the xCELLigence DP
Price on request
CIM Plate 16 for the xCELLigence DP 1×6 Plates
E-Plate 384 for the xCELLigence HT
Price on request
E-Plate 384 for the xCELLigence HT. 2×5 Plates
E-Plate 16 VIEW for the xCELLigence DP
Price on request
E-Plate 16 VIEW for the xCELLigence DP 1×6 Plates
E-Plate 96 VIEW for the xCELLigence SP/MP
Price on request
E-Plate 96 VIEW for the xCELLigence SP/MP 1×6 Plates
E-Plate CARDIO 96 for the xCELLigence cardio
Price on request
E-Plate CARDIO 96 for the xCELLigence cardio 1×6 Plates
E-Plate L8 for the iCELLigence
Price on request
E-Plate L8 for the iCELLigence 1×6 Plates
E-Plate L8 PET for the iCELLigence
Price on request
E-Plate L8 PET for the iCELLigence 1×6 Plates
E-Plate 16 PET for the xCELLigence DP
Price on request
E-Plate 16 PET for the xCELLigence DP 1×6 Plates
E-Plate 96 PET for the xCELLigence SP/MP
Price on request
E-Plate 96 PET for the xCELLigence SP/MP 1×6 Plates
E-Plate cardioECR 48 for the xCELLigence cardioECR
Price on request
E-Plate cardioECR 48 for the xCELLigence cardioECR 1×6 Plates
E-Plate Insert 16 for iCELLigence and xCELLigence
Price on request
E-Plate Insert 16 for iCELLigence and xCELLigence
RTCA E-Plate Insert 96
Price on request
RTCA E-Plate Insert 96
Get in touch
Dr. Kathrin Ringwald
Sales
 Your Partner in Cell Research
Your Partner in Cell Research
Karl-Ferdinand-Braun-Straße 2
28359 Bremen, Germany

OMNI Life Science GmbH
Laufenstraße 90
4053 Basel, Switzerland














