Overview and options
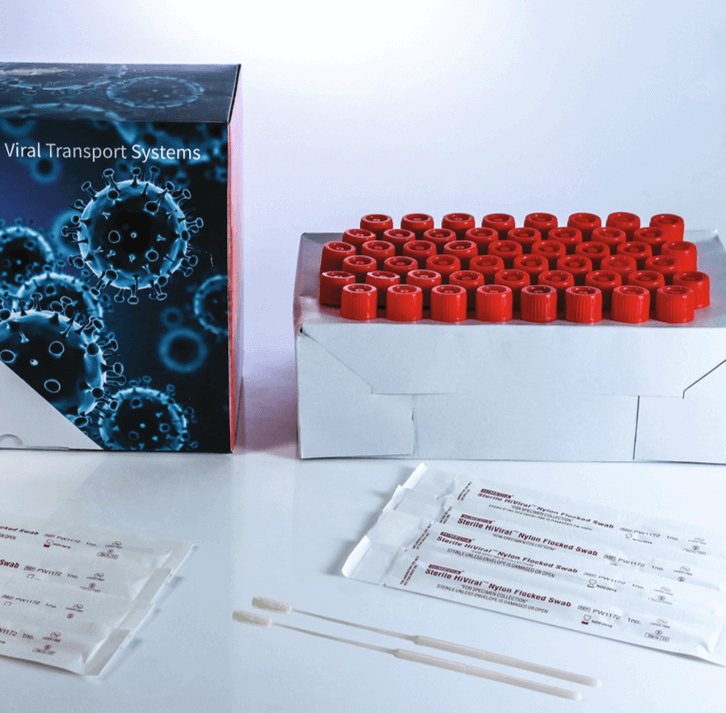
Virus Transport Media are available in several standard configurations. Alternatively, we provide a step-by-step VTM configurator to design VTM Kits as needed.
Testing for SARS-CoV-2/COVID 19 is a major challenge currently and it begins with sample collection and transport. Different testing laboratories however require different combination of swabs, tubes with or without media.
Tubes
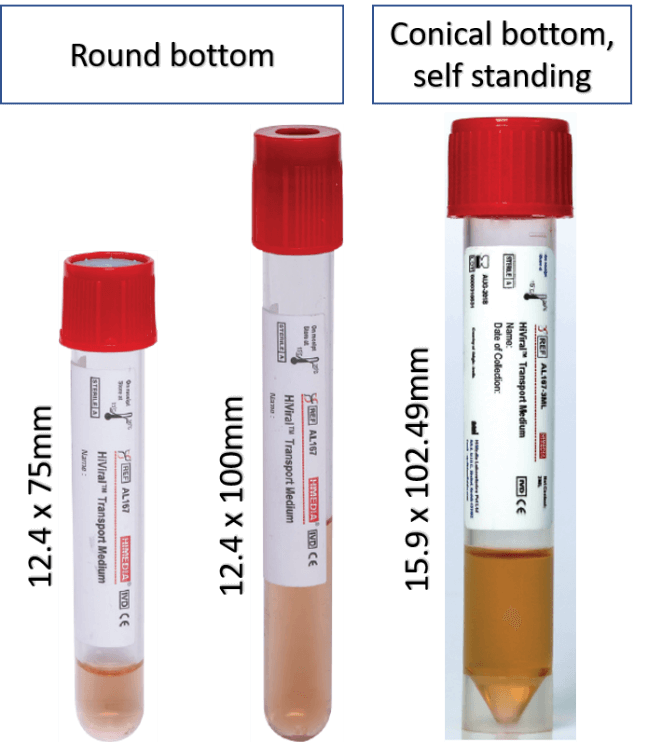
3 versions of standard tubes are available. Two types with round bottom vary in length. Alternatively, there is a self-standing tube with conical bottom available. The VTM Configurator allows request for alternative designs.
COVIDSafeTM - inactivate and be safe
Often, nucleic acid (NA) tests do not require viable viruses but intact nucleic acid particles of the viral genome.
has been designed to support majority of currently approved SARSCoV-2/COVID-19 tests in the world are molecular based tests like RT-qPCR.

provides
Safety – On-the-spot inactivation of samples minimizes such risks of accidental spread of disease.
Ease of handling – Due to inactivation, sample virulence is lost and samples can be processed under Biosafety Level II.
Easy and economical transport – Samples can be stored and shipped at ambient temperature, thereby, eliminating the need of cold chain maintenance and reducing shipping/ packing costs.
Storage and shelf life – Store at room temperature, away from bright light. Use before date of expiry given on the product label.
comes either in 1.5ml or 3ml in self standing tubes with or without Nasopharyngeal Nylon Flocked Swab
HiViralTM Transport Medium
Transport medium is a specially formulated medium for collection, transport and long term freeze storage of viruses. It is designed to maintain the optimum viability and virulence of the viral sample.
HiMedia and
Transport Medium Product code AL167 are also listed at FDA for companies that have validated and intend to distribute VTM as set forth in Section IV.B of the FDA’s COVID-19 Transport Media Policy.
Transport Medium is made of Hanks Balanced Salt Solution and contains a protective protein antibiotic to control microbial contamination and buffers to control the pH. Phenol red is used as a pH indicator. The medium also contains a cryoprotectant which helps in preserving the viruses, if specimens are frozen for prolonged storage.
comes either in 1.5ml or 3ml in self standing tubes with or without Nasopharyngeal Nylon Flocked Swab
Swabs

Different options for swabs are available
- Nylon Swabs
- Nasopharyngeal and Oropharyngeal
- Oropharyngeal Viscose Swab with different beak points (30, 65 and 80mm)
Swabs are also available separately or other swabs are available on request.
VTM Standard Configurations
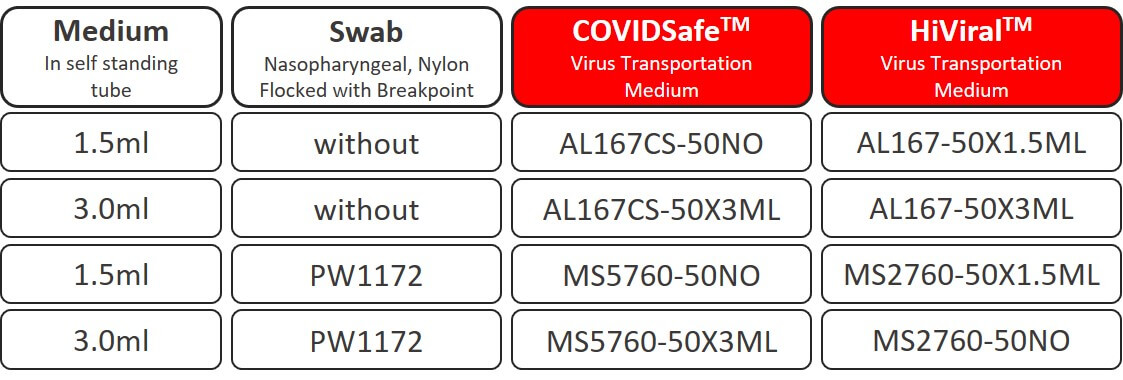
A selection of standard configurations is available for short term sampling the image below provides a summary of the most common combinations:
Virus Inactivation Medium with Virus Inactivation Medium in self-standing tube
- AL167CS-50NO – 1.5ml Virus Inactivation Medium in self-standing tube (50 Pcs)
- AL167CS-50X3ML – 3ml Virus Inactivation Medium in self-standing tube (50 Pcs)
Virus Inactivation Kit with Virus Inactivation Medium in self-standing tube and one Sterile Nasopharyngeal Nylon Flocked Swab with Breakpoint
- MS5760-50NO – 1.5ml Virus Inactivation Medium in self-standing tube and Sterile Nasopharyngeal Nylon Flocked Swab with Breakpoint (50 Pcs)
- MS5760-50X3ML – 3ml Virus Inactivation Medium in self-standing tube and Sterile Nasopharyngeal Nylon Flocked Swab with Breakpoint (50 Pcs)
Transport Medium with Viral Transport Medium in self-standing tube
- AL167-50X1.5ML – 1.5ml Virus Transport Medium in self-standing tube (50 Pcs)
- AL167-50X3ML – 3ml Virus Transport Medium in self-standing tube (50 Pcs)
Transport Kit with Viral Transport Medium in self-standing tube and one Sterile Nasopharyngeal Nylon Flocked Swab with Breakpoint
- MS2760-50X1.5ML – 1.5ml Virus Transport Medium in self-standing tube and Sterile Nasopharyngeal Nylon Flocked Swab with Breakpoint (50 Pcs)
- MS2760-50NO – 3ml Virus Transport Medium in self-standing tube and Sterile Nasopharyngeal Nylon Flocked Swab with Breakpoint (50 Pcs)
Nasopharyngeal Nylon Flocked Swab with Breakpoint
- PW1172 –
Transport Consumable Kit with Empty self-standing tube and sterile Nasopharyngeal Nylon Flocked swab with Breakpoint
- TCP297 – Empty self-standing tube and sterile Nasopharyngeal Nylon Flocked swab with Breakpoint
For more details look at ordering information or download additional information in download section.
VTM Configurator
Here you either have the option to select one of our standard VTM configurations or define your customized VTM kit by simply fill out some fields:
Downloads
Product Information
HiViralTM Transport Medium With Viral Transport Medium
Recommended for collection and transport of viruses
Technical Datasheet
COVIDSafeTM Virus Inactivation Medium
Product Information
HiViralTM Transport Kit With Viral Transport Medium in self-standing / conical bottom tube and One Sterile Nasopharyngeal Nylon Flocked Swab
Product Information
COVIDSafeTM Virus Inactivation Kit
Product Information
HiViralTM Nasopharyngeal Nylon Flocked Swab
Get in touch
Dr. Joachim Pavel
Sales
 Your Partner in Cell Research
Your Partner in Cell Research
Karl-Ferdinand-Braun-Straße 2
28359 Bremen, Germany

OMNI Life Science GmbH
Laufenstraße 90
4053 Basel, Switzerland